High-altitude mountaineering induces adaptive gut microbiome shifts associated with dietary intake and performance markers
Microbiome and nutritional adaptations to High-Altitude stress
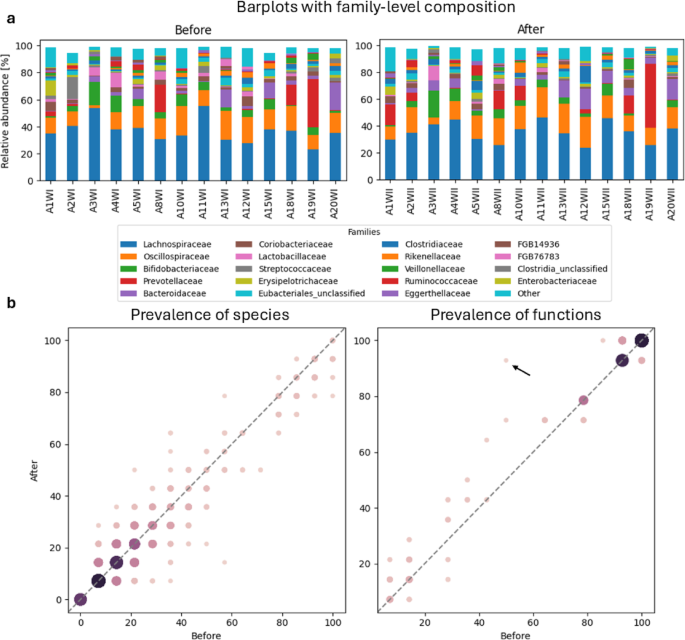
The observed changes in gut microbiome compositions before and after the high-altitude mountaineering expedition indicate adaptive shifts in the gut microbiome and host physiology in response to extreme environmental and physiological stressors. Our findings demonstrate that exposure to low atmospheric pressure, hypoxic conditions, and challenging dietary changes can reshape the gut microbiome, potentially affecting host metabolism, immune function, and overall performance. Similar to previous studies showing that altitude-induced hypoxia alters energy metabolism and physiological adaptations in athletes35,36,37, we identified functional shifts in the gut microbiome, such as increased prevalence of the glucose and glucose-1-phosphate degradation pathway, which was present in half of individuals before the expedition and in almost all participants upon their return. In addition, the abundance of this pathway increased significantly. The presence of this pathway likely reflects not only the elevated intake of simple carbohydrates during the expedition – through consumption of energy gels, sweetened bars, isotonic drinks, and freeze-dried meals38,39 – but also an adaptive microbial response to environmental and physiological stressors, including hypoxia and increased energy demand. While the specific glucose and glucose‑1‑phosphate degradation pathway has not been directly reported in earlier hypoxia studies, a number of investigations in altitude-adapted populations suggest broader functional shifts in microbial carbohydrate metabolism under environmental stress40,41,42. The observed increase in the prevalence of the glucose and glucose-1-phosphate degradation pathway may indicate both an increase in substrate availability and a microbiome strategy to maximize ATP yields in the presence of restricted oxygen.
From a nutritional perspective, our results highlight the importance of vitamin intake, particularly vitamins C and B6, in modulating microbial metabolic pathways such as the preQ0 biosynthesis pathway. The observed association between the preQ0 biosynthesis pathway and dietary intake of vitamins B6 and C prior to the high-altitude mountaineering expedition highlights the intricate interplay between nutrition, microbiome dynamics, and metabolic adaptation. Vitamin B6, known for its role as a cofactor in amino acid metabolism43, is essential for the synthesis of nucleotides and neurotransmitters. This vitamin may enhance the efficiency of metabolic processes involved in preQ0 production, as the pathway is likely reliant on amino acid precursors. Additionally, vitamin C serves as a potent antioxidant, crucial for mitigating oxidative stress that can be exacerbated in high-altitude environments44,45,46. Its antioxidant properties may also support beneficial gut microbial populations capable of metabolizing this vitamin and subsequently influencing pathways like the preQ0 biosynthesis pathway. The dietary intake of these vitamins may modulate gut microbiome composition, ensuring the availability of substrates necessary for microbial metabolism, which in turn supports the preQ0 biosynthesis pathway. This association underscores the significance of adequate vitamin intake in preparing the body for the metabolic demands of high-altitude exposure, where increased energy expenditure and altered oxygen availability necessitate robust cellular adaptations47.
An analysis of Bray-Curtis distances between baseline and post-expedition samples revealed varying degrees of microbiome alterations among the participants. Individuals with greater microbiome shifts responded to the high-altitude conditions better through improvements in fitness markers such as a higher pace at the VT2. This observation suggests that greater microbiome plasticity might be associated with improved metabolic or cardiorespiratory adaptation under environmental stress, such as hypobaric hypoxia19,48. Interestingly, participants with larger microbiome shifts also had significantly richer and more diverse baseline microbiomes. Greater microbial diversity has been previously linked with enhanced resilience and adaptive capacity of the gut ecosystem, potentially allowing for more dynamic functional reorganization in response to stressors like high altitude, changes in diet, or physical exertion49,50,51. This was accompanied by insignificant but notably higher intakes of macronutrients, such as manganese, calcium, vitamin A, magnesium and phosphorus before the expedition, which have been implicated in supporting microbial diversity, intestinal barrier integrity, and immune function52,53,54,55,56. Finally, their microbiomes were enriched in Clostridium fessum, Streptococcus salivarius and the inosine 5’-phosphate degradation pathway. S. salivarius has been identified as a commensal species with immunomodulatory properties and the ability to produce bacteriocins and anti-inflammatory compounds57,58, while Clostridium fessum may contribute to short-chain fatty acid production, although its role remains less well understood. SCFAs (e.g., acetate, propionate, butyrate) are key microbial metabolites derived from fermentation of dietary fibers and play important roles in host energy metabolism, immune regulation, and gut barrier integrity59. The presence of the inosine 5’-phosphate degradation pathway may reflect increased nucleotide turnover and energy metabolism potential, which has been observed in microbiomes adapting to metabolic stress60. These preliminary findings suggest that baseline microbiome composition and its capacity to dynamically respond to environmental stressors may influence the physiological outcomes of high-altitude exposure. Future studies with larger cohorts are needed to explore these associations further and confirm causality. A similar performance-oriented plasticity is emphasized in our recent narrative review of athletic gut microbiomes61, which shows that elite performers consistently harbour SCFA- and bile-acid-producing consortia that safeguard energy supply, immunity and barrier function during heat, hypoxia and prolonged exertion – paralleling the altitude-driven shifts observed here.
Importantly, several microbial features that differed within our cohort showed significant correlations with physiological read-outs, suggesting that the compositional shifts we observed may have functional – rather than purely cosmetic – consequences. Before the expedition, several microbiome-host links converged on the same theme: a Prevotella-rich, rhamnose-active community was associated with superior lower-limb power and tighter electrolyte control. Specifically, the dTDP-β-L-rhamnose-biosynthesis pathway showed a strong positive correlation with both total work and mean anaerobic power of the lower limbs (ρ = 0.96 for each; Table 2), while serum potassium related positively to Prevotella copri (ρ = 0.98) and negatively to Bacteroides uniformis (ρ = −0.97). Taken together, these baseline patterns indicate that athletes entering the expedition with a Prevotella-rich enterotype may enjoy more efficient mineral absorption and rapid carbohydrate utilisation19 – an interpretation consistent with endurance cohorts in which Prevotella enrichment tracks high-carbohydrate, fibre-dense diets and long weekly training volumes62. In our climbers, the same triad – P. copri, elevated potassium, and the rhamnose nucleotide-sugar route – suggests a microbiome “fitness reserve” that supports energy flux and electrolyte stability when oxygen becomes limited at altitude.
Consistent with the idea that pre-existing gut configurations modulate later physiological responses, our recent case-control study in collegiate men performing maximal Wingate and Bruce tests revealed a different, but conceptually similar, pattern: baseline enrichment of Clostridium phoceensis and Catenibacterium spp. predicted blunted IL-1α and TIMP-1 responses to the aerobic (Bruce) protocol, whereas anaerobic testing elicited distinct microbiome–cytokine couplings driven by ethanolamine- and pyrimidine-related pathways28. After the climb, a distinct post-expedition signature emerged. Enrichment of the NAD+de-novo-biosynthesis I pathway correlated positively with circulating calcium (ρ = 0.99) and with basophil counts (ρ = 0.96), whereas the super-pathway of L-serine/glycine biosynthesis related inversely to sodium (ρ = − 0.96) but positively to HDL cholesterol (ρ = 0.96). In parallel, the myo-,chiro- and scyllo-inositol degradation pathway also tracked with basophils (ρ = 0.96), and guanosine-ribonucleotide biosynthesis showed an inverse relationship with mean platelet volume (ρ = − 0.96). Post-expedition functional read-outs therefore realigned earlier associations: at baseline both guanosine-ribonucleotide biosynthesis and the myo-/chiro-/scyllo-inositol degradation pathway tracked positively with serum sodium, whereas after altitude exposure the same routes coupled to MPV and basophils, respectively. This directional reversal underscores a flexible microbiome contribution to nutrient flux, electrolyte balance and immune signalling under chronic hypoxia. Taken together, these links connect post-expedition shifts in nucleotide, redox-cofactor and amino-acid metabolism to haematological (basophils, MPV), electrolyte (sodium, calcium) and lipid (HDL) read-outs, supporting a role for the gut microbiome in coordinating metabolic and immune adaptation under prolonged hypoxia and high energy demand63. Importantly, intestinal-permeability markers (zonulin and α₁-antitrypsin) remained unchanged, consistent with recent reports that well-acclimatized alpinists can maintain epithelial integrity under hypobaric stress8.
A differential enrichment analysis with LEfSe in our study identified 3 species, namely Blautia luti, Romboutsia timonensis and GGB4569_SGB6310, enriched in abundance after the expedition. Similarly, Su et al. showed a relative and absolute increase in Blautia A abundance in men who were exposed to high altitudes (> 3,600 MASL) for more than 2 months41. Blautia A species were shown to be involved in a module of “cobalamin biosynthesis” and butyric acid production that are both beneficial to microbial ecosystems and intestinal epithelial cells41,64. An in vivo animal experiment supported a crucial role of Blautia A species in facilitating host fitness to hypoxia environments, likely via anti-inflammation and intestinal barrier protection to maintain intestinal health, thereby suggesting a high translational potential of Blautia A species as a candidate probiotic agent for the prevention or treatment of hypoxia-associated maladaptation or disorders. Taken together, these observations of increased abundance of Blautia A imply this genus is a beneficial high-altitude bacterial group that may play an important role in promoting acclimatization and adaptation to high-altitude conditions41.
We observed significant microbial functional associations with dietary intake after the expedition. Notably, strong positive correlations with coenzyme A biosynthesis and various dietary components suggest that these nutrients may enhance microbial metabolic pathways essential for energy production and lipid metabolism. Similarly, positive associations of L-arginine biosynthesis with overall carbohydrate intake and the assimilation of carbohydrates indicate that dietary carbohydrates may support microbial synthesis of this amino acid, which is crucial for various physiological functions18,19,63. Furthermore, associations of the pentose phosphate pathway with selenium and plant protein with sucrose biosynthesis highlight the potential role of specific micronutrients and macronutrients in modulating microbial metabolic pathways. On the other hand, negative associations of adenine and adenosine salvage pathways with sodium intake, as well as between L-rhamnose biosynthesis and animal protein consumption could suggest competitive inhibition or substrate limitation in these metabolic processes. Overall, the results emphasize complex interactions between dietary components and microbial metabolic activities in the gut. However, the sample size for this analysis was limited (n = 7); thus, future validation of these findings in a larger cohort is necessary.
Practical Implications, Limitations, and future perspectives
From an applied perspective, these findings provide valuable guidance for mountaineers, coaches, and sports nutritionists. Pre-expedition strategies might include optimizing micronutrient status, particularly for vitamins and minerals shown here to correlate with beneficial microbial functions and performance markers. During expeditions, integrating nutrient-dense, microbiome-supportive foods – even if partially in the form of supplements or freeze-dried products – may help maintain beneficial gut microbiota profiles and associated metabolic functions. Beyond the athletic context, our findings contribute to a growing body of literature on the role of gut microbiome in adaptation to extreme environments40,41,65,66,67,68,69, highlighting opportunities to leverage nutritional interventions to support both health and performance.
Climbers should avoid taking non-prescribed antibiotics when possible, as agents such as nifuroxazide can temporarily reduce gut microbial diversity. Gradual acclimatization, along with a diet rich in fiber, polyphenols, and adequate carbohydrates, may further support gut barrier integrity and mitigate AMS-related inflammation61.
Future research should aim to confirm these findings in larger cohorts and explore targeted interventions-such as prebiotics, probiotics, postbiotics, or tailored micronutrient supplementation to promote a beneficial gut microbiome response in high-altitude environments. Integrating metagenomics with host transcriptomics, metabolomics, and immune profiling, as well as conducting controlled feeding studies, could further elucidate the mechanisms linking the gut microbiome, diet, and physiological adaptation in extreme sports and beyond.
Our study has some limitations. The climbers did not constitute a single group, led by a single climbing goal in the territory of a single country and mountain range. Climbers, characterized by the characteristics described in the study, constitute a small but elite group in Poland, hence several distinct expedition groups, climbing at similar altitudes and with similar sporting goals, but in different parts of the world, qualified for the study. Gathering and encouraging participants was one of the most challenging stages of the project. While all expeditions involved high-altitude exposure above 3000 MASL, other factors such as temperature, humidity, and radiation levels were not controlled for and may have contributed to interindividual differences in microbiome responses. Future studies conducted in more standardized settings are warranted to disentangle the specific environmental drivers of microbiome changes at altitude.
Due to the small number of GI disturbance (n = 3) and AMS (n = 1) events, the study lacked the power to detect reliable microbiome correlates. Exploratory diversity and taxonomic scans did not reveal outliers (see Supplementary Fig. 3). Likewise, exploratory alpha- and beta-diversity metrics and LEfSe screening (species and MetaCyc pathways, p < 0.05) failed to identify discriminant features. Larger cohorts are needed to clarify any causal links.
In addition to the level of hypoxia, other factors such as nutrition and the type of physical activity performed in the mountains, as well as the number of active days versus the number of rest days, may contribute significantly to the observed changes in microbiome. Climbing groups active in Peru could have used the downtime between several days of mountain activity to descend to a town at the foot of the mountains to compensate for the negative energy balance there, while expedition groups in Kyrgyzstan, Nepal or Pakistan were dependent on the food they took into the mountains or that the expedition agency offered them.
We acknowledge that some participants consumed sports supplements and functional foods for athletes, which – according to Álvarez-Herms et al.23 – may influence gut microbiota composition and gut barrier integrity in athletes. Although supplement intake was recorded in our study, we did not specifically analyze the potential impact of additives commonly found in these products (e.g., emulsifiers, artificial sweeteners, preservatives) on the gut microbiota. Furthermore, we did not observe a clear association between general supplement use and microbiome shifts or performance outcomes. Nevertheless, considering the small sample size, individual variability, and heterogeneous supplement composition, we cannot fully exclude a modulatory effect. Future studies should incorporate detailed assessments of supplement ingredients, including non-nutritive additives, to better understand their influence under high-altitude stress conditions.
link






